Update: October 8, 2021
Consistent with earlier two-dose findings, the most commonly reported local reactions after a third COVID-19 mRNA vaccine (Moderna or Pfizer) among blood cancer patients were sore arm (57%) and injection site pain (18%). The most common systemic reactions were fatigue (31%), muscles aches/pain (18%) and headache (17%). Allergic reactions including rash and shortness of breath were rare, occurring in less than 1% of those vaccinated.
These results are based on patient reports from more than 2,400 participants in the LLS National Patient Registry who received a third dose of either Moderna or Pfizer COVID-19 vaccines between August 13, 2021 and September 20, 2021.
“Most reactions to these vaccines—including a third dose—are mild and temporary,” said LLS Chief Medical Officer Dr. Gwen Nichols. “I urge all blood cancer patients and survivors to get vaccinated—including with the third dose, unless told otherwise by their doctor.”
LLS recently reported that most blood cancer patients benefit from an additional COVID-19 vaccine dose. Read more here.
The CDC advises using the same vaccine for all three doses, but if the original vaccine is not available or unknown, either vaccine can be given. The minimum wait time between the second and third dose is 28 days. Blood cancer patients and survivors should get the third dose as soon after 28 days as possible.
(Originally published April 23, 2021)
Background and Purpose
Because of the health emergency created by the COVID-19 virus, three COVID-19 vaccinations have been authorized by the U.S. FDA for Emergency Use Administration (EUA). The vaccine clinical trials were streamlined and essentially excluded people with a current or past diagnosis of cancer. Therefore, how people with blood cancer respond to the COVID-19 vaccinations is unknown.
The Leukemia & Lymphoma Society National Patient Registry, created in honor of Michael Garil, is collecting data to provide more specific information about how people with blood cancer respond to COVID-19 virus and/or COVID-19 vaccination. The Registry includes patients and survivors with every type of blood cancer; participants include those on active treatment, post-treatment or in a “watch and wait” period (“treatment naïve”).
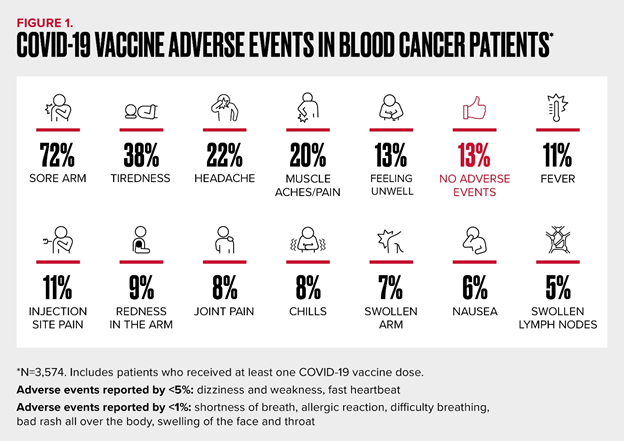
This preliminary report includes vaccine adverse events data from 3,574 Registry participants who had at least one dose of any currently authorized COVID-19 vaccine (BNT162b2 from Pfizer and BioNTech, mRNA-1273 from Moderna, JNJ-78436735 from Janssen Pharmaceuticals Companies of Johnson & Johnson). Every patient who was planning to be vaccinated was also offered the opportunity to provide blood samples to test for the COVID-19 spike and neutralizing antibody. Data about the subset of patients who agreed to participate will be available by summer 2021. (Update: press release now available here).
Results
Between February 22 and April 8, 2021, 4,505 blood cancer patients and survivors (average age 64 years) completed a survey about their experiences with COVID-19 and vaccination. Of those, 3,574 had a least one dose of the vaccine and reported their experience with adverse events.
The most common diagnoses among respondents were chronic lymphocytic leukemia, non-Hodgkin lymphoma, Waldenström macroglobulinemia and myeloma. Smaller numbers of patients were diagnosed with myelodysplastic syndromes, chronic myeloid leukemia, myeloproliferative neoplasms and acute leukemias.
Most respondents received either the Pfizer (53%) or Moderna (44%) vaccine; 2% received JNJ and less than one percent received AstraZeneca (not currently authorized for use in the U.S.) or did not indicate a vaccine. Data are reported as an aggregate that include all vaccines.
Patient-reported adverse events in the Registry closely mirrored those seen in the large randomized, controlled clinical studies of healthy volunteers, 56 years of age and older.1,2
Consistent with the trials, the most commonly reported local reactions were sore arm (72%) and injection site pain (11%) (Figure 1). The most commonly reported systemic reactions were fatigue (38%), headache (22%) and muscle aches (20%), followed by feeling unwell (13%) and fever (11%). More than 1 in 10 (13%) reported no local or systemic reactions and less than 1% did not know or could not recall. Other important, but less common side effects included chills (8%), nausea (6%) and lymphadenopathy (5%). Allergic reactions including rash and shortness of breath were rare (<1%).
Discussion
These are the first data reported from the LLS National Patient Registry COVID-19 project. While collection of analysis of information is ongoing this “real world” survey of patients and survivors, provides important insights from blood cancer patients and survivors who have been safely vaccinated. This can help reassure those who have not yet been vaccinated and give healthcare professionals important initial safety data specific to this at-risk population.
While the side effect profile of the currently authorized vaccines appears to be very similar in blood cancer patients/survivors compared to the general public, reports of lymphadenopathy among 1 in 20 vaccine recipients may be of concern for blood cancer patients and their providers. Many patients with blood cancer are followed for lymph node enlargement as evidence of their tumor returning. If the lymph node enlargement is near the injection site, and disappears, it is likely due to the vaccination. However, any change in lymph nodes in a blood cancer patient should be discussed with the patient’s oncology healthcare provider.
Reassurance about vaccine safety will be important because a recent survey of more than 6,000 blood cancer patients carried out by LLS, Boston University Questrom School of Business and The Behaviouralist, a London-based research consultancy, showed that nearly one in five blood cancer patients are hesitant about receiving the COVID-19 vaccine. The most common reasons for vaccine hesitancy were concerns about side effects and a belief that vaccines have not been tested properly.
While these preliminary data appear to confirm that vaccine safety and tolerability are similar in blood cancer patients and survivors compared to those with no history of cancer, important questions still need to be answered about how well these vaccines induce immunity in these patients. The LLS National Registry is currently collecting these important data. In the meantime, for a disease as deadly as COVID-19, any protection from vaccines is better than no protection, especially for blood cancer patients who are at much increased risk of complications and death from COVID-19 infection than those without cancer.3-6
For more information, check out our COVID-19 Resource Page
REFERENCES
1. Polack FP, Thomas SJ, Kitchin N, et al., Safety and efficacy of the BNT162b2 mRNA Covid-19 vaccine. N Engl J Med. 2020; 383 (27):2603-2615.
2. Baden LR, El Sahly HM, Essink B, et al. Efficacy and safety of the mRNA-1273 SARS-CoV-2 vaccine. N Engl J Med. 2021; 384(5): 403-416.
3. Metha V, Goel S, Kabarriti, R. Case fatality rate of cancer patients with COVID-19 in a New York hospital system. Cancer Disc. 2020; 10(7): 935-941.
4. Vijenthira A, Gong IY, Fox TA, et al. Outcomes of patients with hematologic malignancies and COVID-19: a systematic review and meta-analysis of 3377 patients. Blood. 2020;136(25):2881-2891.
5. Lee LYW, Cazier J-B, Starkey T et al. COVID-19 prevalence and mortality in patients with cancer and the effect of primary tumor subtype and patient demographics: a prospective study. Lancet Oncol. 2021; 21: 1309-1316.
6. Passamonti F, Cattaneo C, Arcaini L, et al. Clinical characteristics and risk factors associated with COVID-19 severity in patients with haematological malignancies in Italy: a retrospective, multicentre, cohort study. Lancet Haematol. 2020;7(10):e737-e745.